 Other atoms, like nuclides and isotopes, are graphically collected in other tables like the tables of nuclides (often called Segrè charts). The periodic table exclusively lists electrically neutral atoms that have an equal number of positively charged protons and negatively charged electrons and puts isotopes (atoms with the same number of protons but different numbers of neutrons) at the same place. The underlying reason for these trends is electron configurations of atoms. Trends run through the periodic table, with nonmetallic character (keeping their own electrons) increasing from left to right across a period, and from down to up across a group, and metallic character (surrendering electrons to other atoms) increasing in the opposite direction. 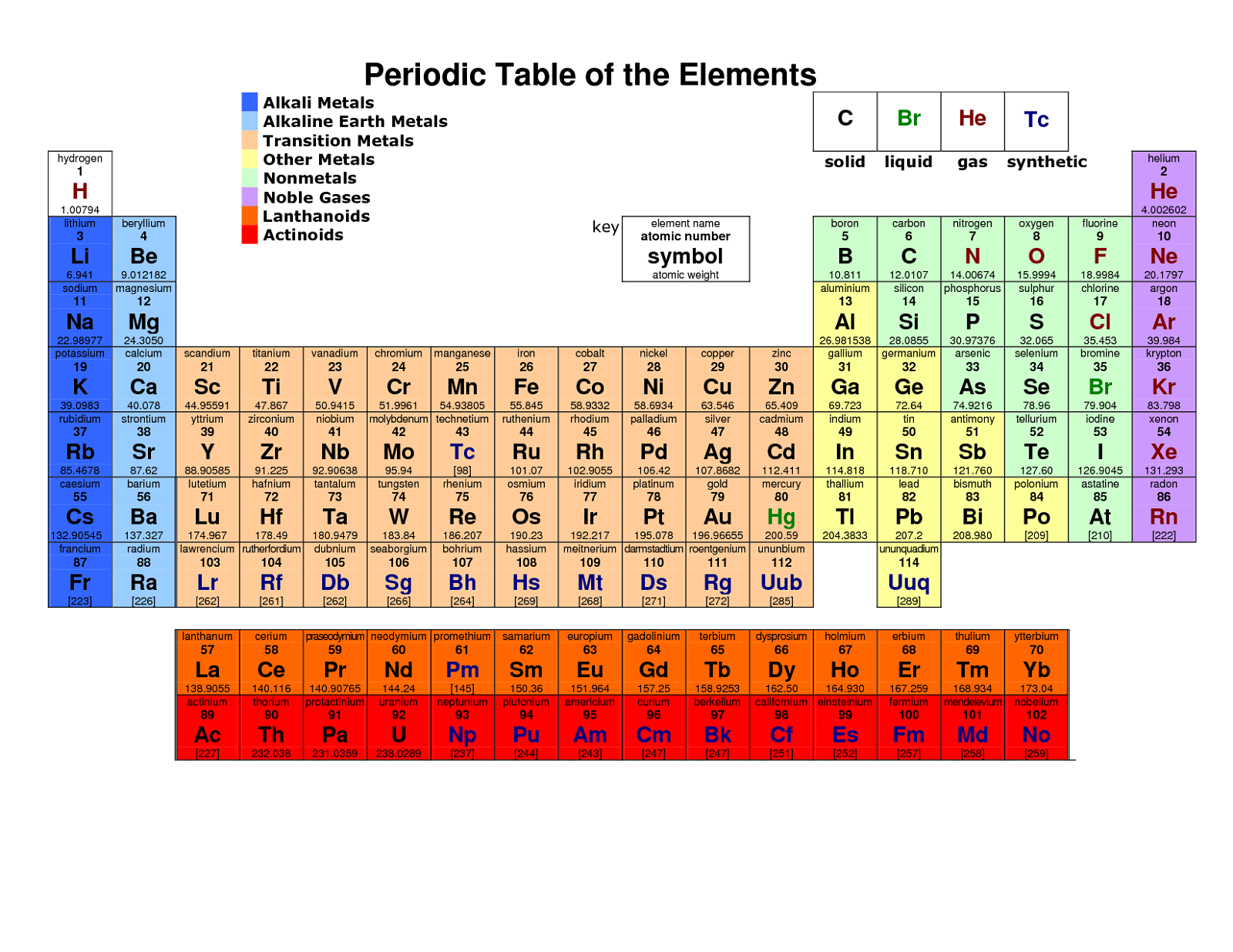
Elements from the same column group of the periodic table show similar chemical characteristics. The rows of the table are called periods, and the columns are called groups. The table is divided into four roughly rectangular areas called blocks. 
It is a graphic formulation of the periodic law, which states that the properties of the chemical elements exhibit an approximate periodic dependence on their atomic numbers. It is widely used in chemistry, physics, and other sciences, and is generally seen as an icon of chemistry. The periodic table, also known as the periodic table of the ( chemical) elements, is a tabular display of the chemical elements. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
