 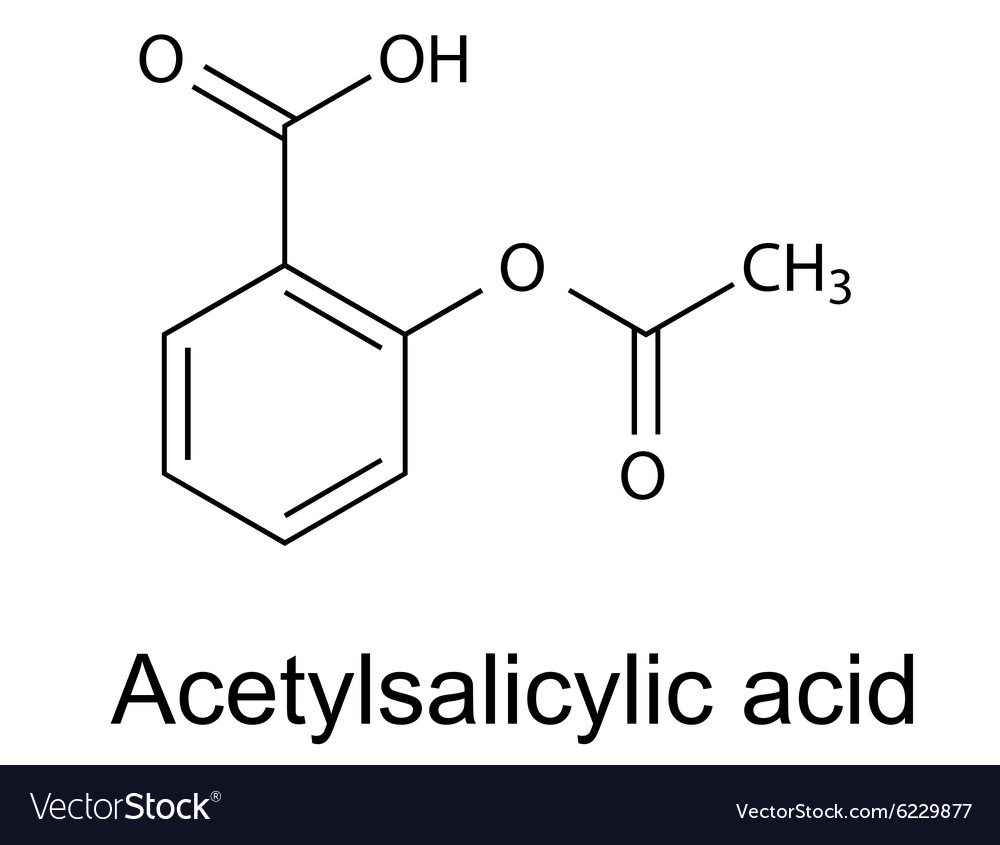 
In addition, alchemilla is also useful for muscle and joint health.Īlchemilla is the subject of more than 90 scientific publications. This plant helps to maintain a good comfort before and during the menstrual cycle. The benefitsĪlchemilla contributes to digestive comfort by promoting the health of the gastrointestinal tract and the well-being of the liver. physical: Salicylic Acid is a white crystalline powder or needle-shaped crystals with sweetish taste soluble in acetone, ether, alcohol, boiling water, benzene and turpentine, sparingly soluble in chloroformbenzene, slightly soluble in water melts at 158C. Moreover, alchemilla helps to fight against pelvic congestion syndrome by reducing the pain it generates.įinally, alchemilla is known for its anti-infectious, antibacterial, antiviral and antifungal properties. Chemical Names: ACETYLSALICYLIC ACID 2-Acetoxybenzoic acid Ecotrin 2- (Acetyloxy)benzoic acid Molecular Formula: C 9 H 8 O 4. It also has vascular properties, in particular thanks to its flavonoids which improve blood circulation. Thanks to its anti-inflammatory, antioxidant, antispasmodic and sedative action, alchemilla offers numerous health benefits. Acetylsalicylic Acid Synthesis By: Elizabeth Renteria Theory. 6 June 2017The preparation of aspirin is conducted through the esterification of salicylic acid and theaddition of acetic. a white crystalline derivative C9H8O4 of salicylic acid used for relief of pain and fever : aspirin See the full definition. Even Tone Facial Serum with Salicylic Acid. How to use acetylsalicylic acid in a sentence. Our concentrated serum with salicylic and glycolic acid removes dead skin cells tp. 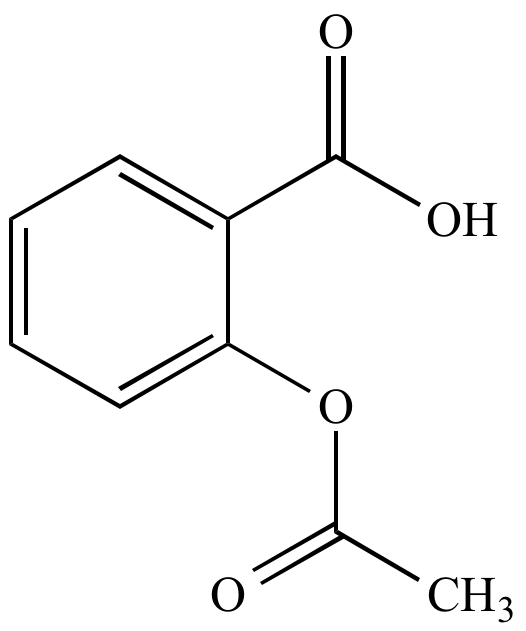 
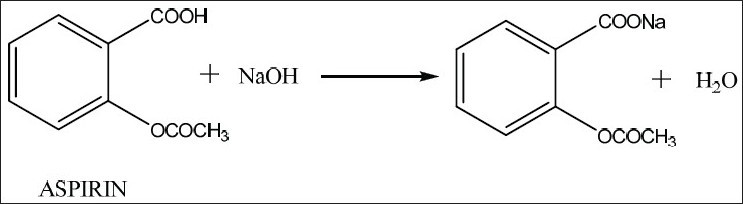
Salicylic acids formula is C6H4(OH)COOH and its IUPAC name is 2-hydroxybenzoic acid. In vivo studies have shown that taking alchemilla reduces endometriosis lesions and the associated inflammatory reactions. The meaning of ACETYLSALICYLIC ACID is a white crystalline derivative C9H8O4 of salicylic acid used for relief of pain and fever : aspirin. In this experiment we will make salicylic acid. The plant contains flavonoids with antioxidant action (quercetol, hyperoside, rutoside) and salicylic acid with anti-inflammatory properties. 1 the carboxyl group on the acetic acid reacts with the hydroxyl group on the salicylic acid to make an ester product of acetylsalicylic acid. He has published extensively in the area of aspirin and prostaglandin research with focus on platelet functions and the clotting system and serves on several national and international review panels and advisory committees on thrombosis research, colorectal cancer and drug development.Indissociable from the alchemists who used to collect the morning dew in the hollow of its leaves, considered as "celestial water" resulting from the condensation of effluvia circulating between the terrestrial and the celestial, the alchemilla has a mystical aspect.īelonging to the Rosaceae family, this plant is also called "women's herb" and has been reputed to cure ailments since the Middle Ages.Ĭontaining tannins (including proanthocyanidols, gallotanins and catechins) with astringent properties and a powerful haemostatic action, alchemilla stops bleeding, particularly in the event of haemorrhage or painful periods, which also enables it to indirectly prevent anaemia.Īlchemilla has many properties. the goal of this experiment is to perform a fischer esterification reaction with salicylic acid and acetic anhydride to synthesize acetylsalicylic acid. Karsten Schror is member of the German National Academy of Science (Leopoldina) and was President of the German Society for Pharmacology from 2005 to 2010. Dissolve 10 g of salicylic acid (o-hydroxybenzoic acid) in dry pyridine (7 ml) in a conical flask of 100 ml. He held guest professorships at the Medical University of South Carolina at Charleston (USA) and the Department of Internal Medicine, Division of Hematology, University of Texas Medical Center, Houston (USA). at Martin-Luther-University Halle-Wittenberg (Germany) and carried out postdoctoral studies at the Universities of Halle, Mainz, Koln, the Wellcome Research Laboratories in Beckenham (UK) and the Jefferson Medical College, Philadelphia (USA). Officially making its highly anticipated return, the Salicylic Acid 2 Solution is back with a formula better than ever. Karsten Schror is Professor of Pharmacology and retired Chairman of the Department of Pharmacology and Clinical Pharmacology at the Heinrich-Heine-University in Dusseldorf, Germany. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
